Ready to streamline healthcare?
Discover how our platform transforms operations. Book a free 30-min consultation to see it in action.




Simplifying Diagnostics. Empowering Your Health.
Access reliable testing information, expert insights, and educational resources to make informed healthcare decisions.


Comprehensive Health Support Platform
Access the trusted resources and expert guidance you need to take control of your health.
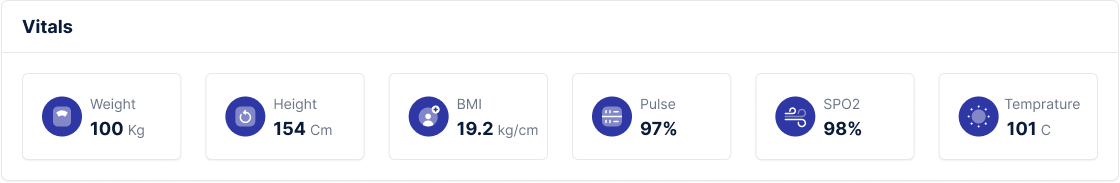
Get clear, detailed explanations for a wide range of medical tests and diagnostic procedures.
Condition overviews, wellness monitoring advice, and step-by-step resources for preventive care.

Understand what certain test results may indicate and explore the next steps for your health journey.
A trusted pathway to qualified healthcare professionals for clarification, consultation, and next-step recommendations.
.png)
Dedicated to Health Literacy and Clarity
Our mission is to make medical information accessible, reduce confusion around diagnostics, and empower every individual.
We provide expert-backed resources and easy-to-digest content to help you navigate your diagnostic journey with confidence.
50+
Comprehensive Health Guides and Topics

Understanding Your Health in 3 Steps
Our platform connects you with the information and experts you need to take informed action.
Find Clear Information
Apply Expert Insights

Talk to a Specialist

Support Tailored for Your Health Journey
Our platform is designed to serve individuals, healthcare providers, and organizations seeking clarity in diagnostics.
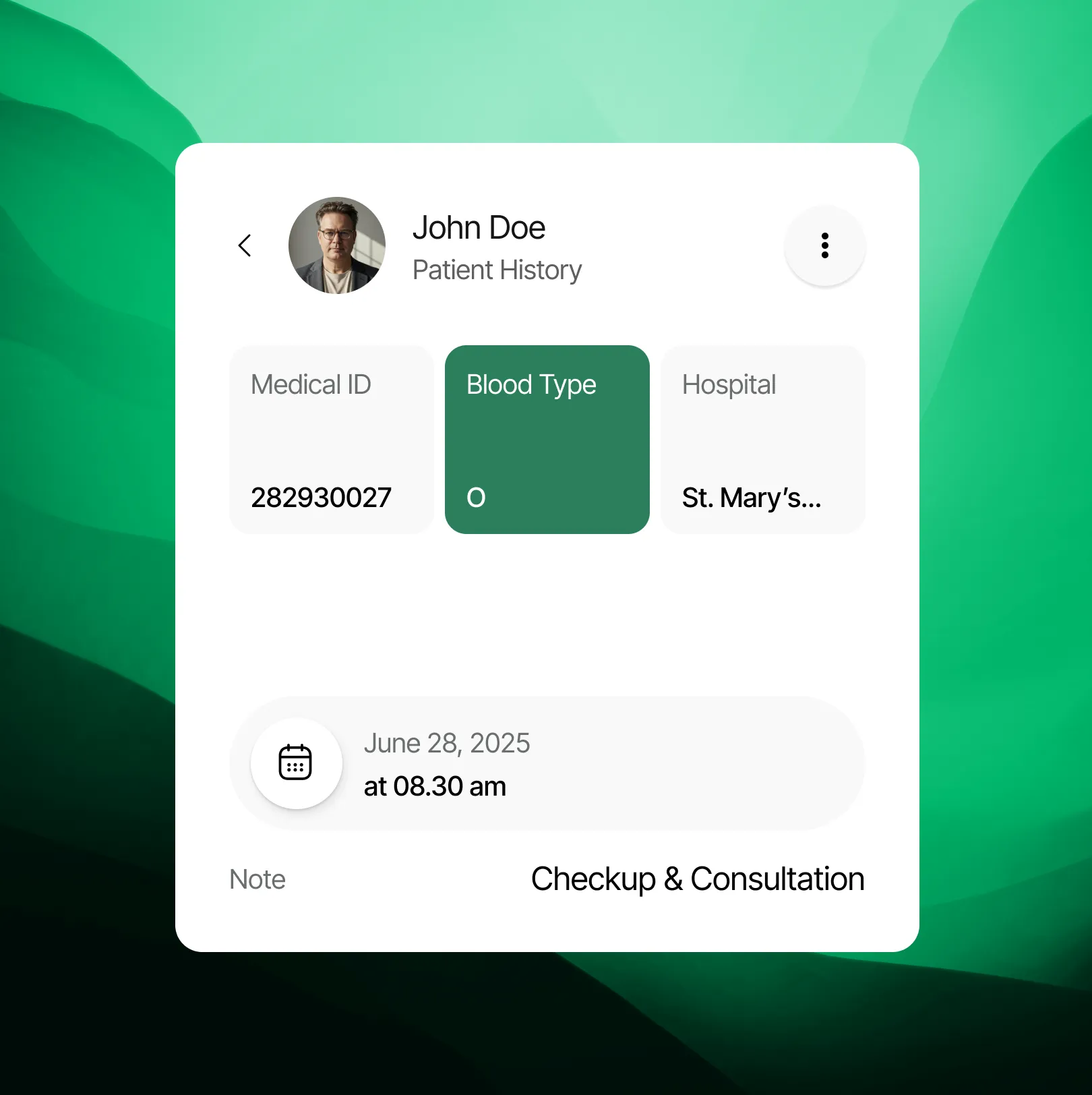
Take control of your health with confidence. Easily access, understand, and interpret testing information and health guides to actively participate in your care and navigate next steps.
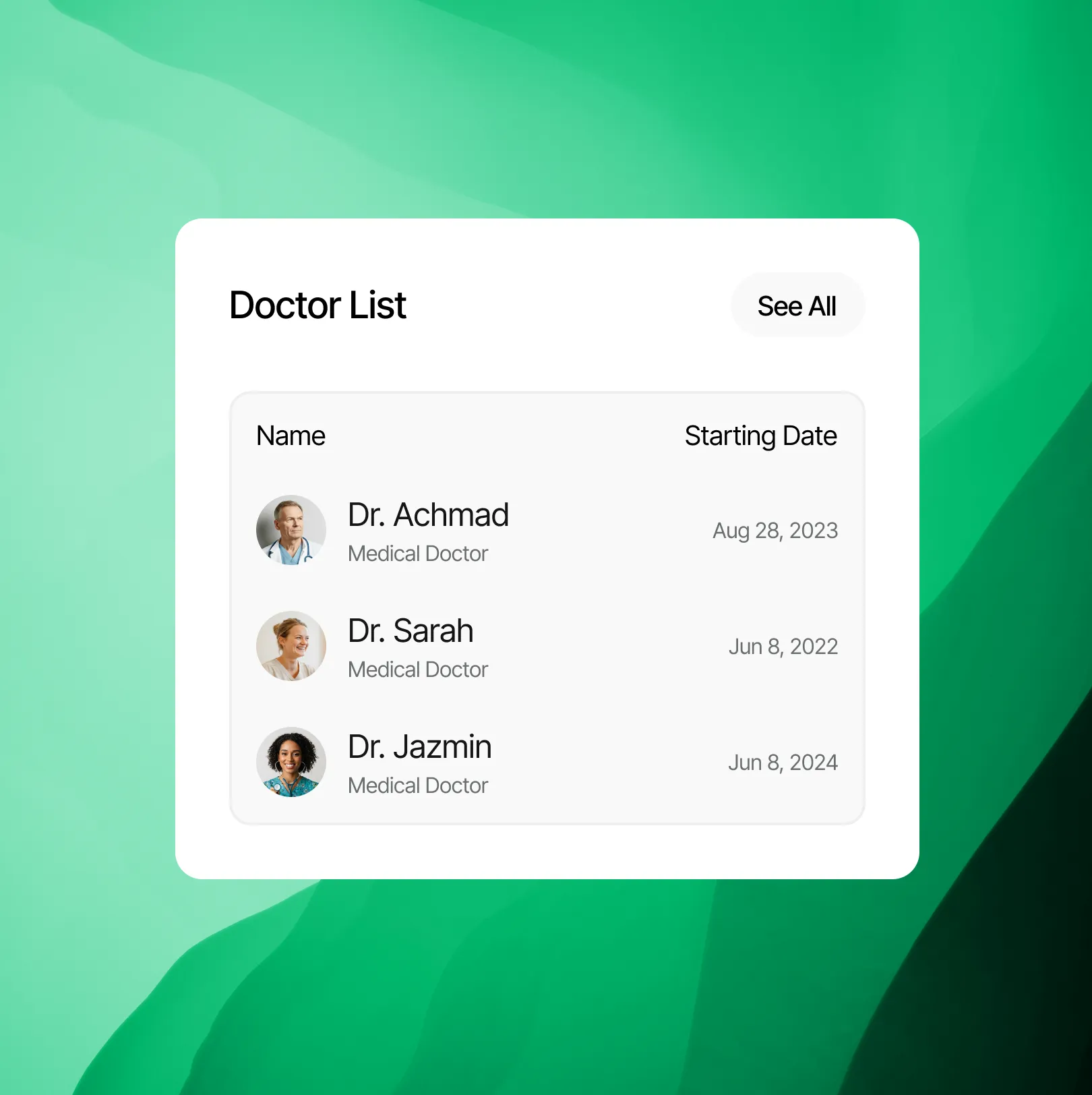
Partner with us to share your expert knowledge. Our platform allows qualified professionals to provide clarification, consultation, and trusted medical guidance to a wide audience seeking diagnostic understanding.
Integrate our comprehensive, expert-reviewed educational resources into your patient support initiatives, employee wellness programs, or community health campaigns to boost general health literacy.
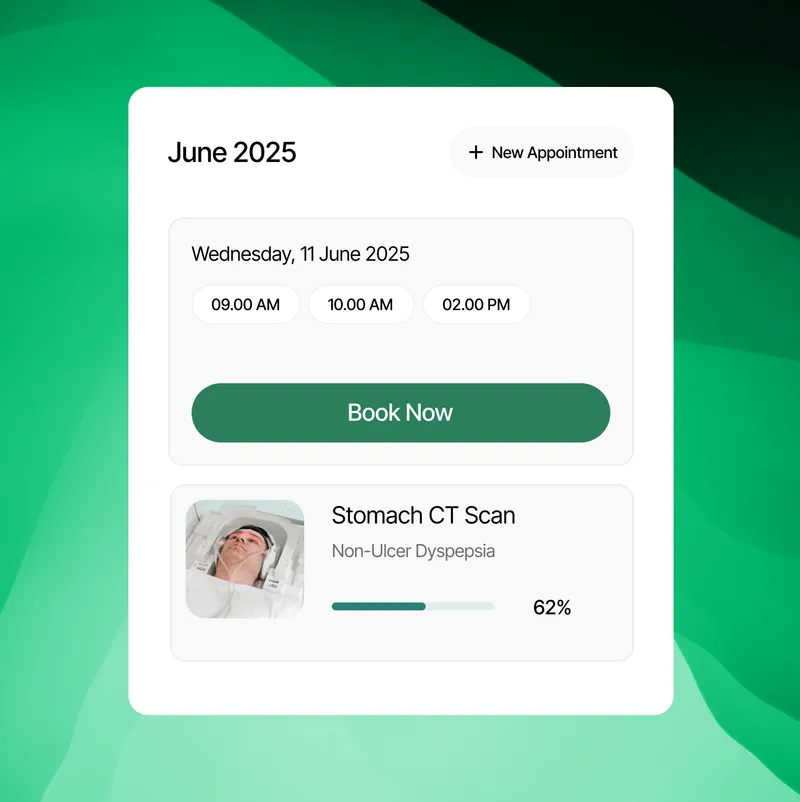
Clarity, Confidence, and Connection
Hear from individuals who have used Group K Diagnostics to understand their health better.
"This platform has completely revolutionized how we manage our campaigns. The data insights are a game-changer for our client reports."
"This platform has completely revolutionized how we manage our campaigns. The data insights are a game-changer for our client reports."
"This platform has completely revolutionized how we manage our campaigns. The data insights are a game-changer for our client reports."
"This platform has completely revolutionized how we manage our campaigns. The data insights are a game-changer for our client reports."
"This platform has completely revolutionized how we manage our campaigns. The data insights are a game-changer for our client reports."
"This platform has completely revolutionized how we manage our campaigns. The data insights are a game-changer for our client reports."
"This platform has completely revolutionized how we manage our campaigns. The data insights are a game-changer for our client reports."
"This platform has completely revolutionized how we manage our campaigns. The data insights are a game-changer for our client reports."
"This platform has completely revolutionized how we manage our campaigns. The data insights are a game-changer for our client reports."
"This platform has completely revolutionized how we manage our campaigns. The data insights are a game-changer for our client reports."
"This platform has completely revolutionized how we manage our campaigns. The data insights are a game-changer for our client reports."
"This platform has completely revolutionized how we manage our campaigns. The data insights are a game-changer for our client reports."
Informed Access & Support
Choose the level of expert guidance and support that best meets your health literacy needs.
- Full Access to Educational Guides
- Jargon-Free Terminology
- Wellness Monitoring Content
- Basic Email Support
- Full Access to Educational Guides
- Jargon-Free Terminology
- Wellness Monitoring Content
- Basic Email Support
Get the Information You Need
Quick answers regarding the platform’s resources, specialists, and data security.
How secure is my medical data?
All data is meticulously encrypted and securely stored using top-tier security standards. This approach not only ensures full privacy for our users but also guarantees compliance with the latest regulations and best practices in data protection.
Who creates and verifies the health guides?
All condition overviews, testing explanations, and health guides are created, written, and continuously reviewed by qualified healthcare professionals and diagnostic specialists to ensure the information is reliable, accurate, and expert-backed.
Can I share the information I find with my doctor?
Yes, absolutely. We encourage you to share our resources with your healthcare provider. Our content is designed to promote an informed discussion between you and your doctor, making your appointments more productive and helping you feel more confident about your care plan.
Is this platform a substitute for official medical advice?
No. Group K Diagnostics provides reliable educational information and access to experts for clarification. It is not a substitute for seeing a qualified healthcare professional, receiving a diagnosis, or obtaining personalized medical advice. Always consult your doctor for medical decisions.
How often are the health and testing guides updated?
Our commitment is to provide the most current information. We continuously monitor changes in medical guidelines, coding (like ICD-10 and CPT codes), and clinical evidence. Most essential content is reviewed and updated annually, with critical changes integrated as soon as new evidence is published.
How do I connect with a diagnostic specialist?
You can use our secure directory to find qualified specialists. The platform provides a clear pathway—usually through secure messaging or virtual consultation booking—to connect with a professional for clarification or next-step recommendations.
Is there a cost to access the educational resources?
Access to the core educational resources, condition overviews, and testing explanations is free for all users. There may be separate costs associated with booking a private consultation with an external specialist through our connection service.
Expert Insights & Wellness Guides
Reliable, easy-to-digest articles on diagnostics, preventive care, and health literacy from our specialists.
Start Your Journey to Health Clarity
Join our SMS list for Exclusive offers!